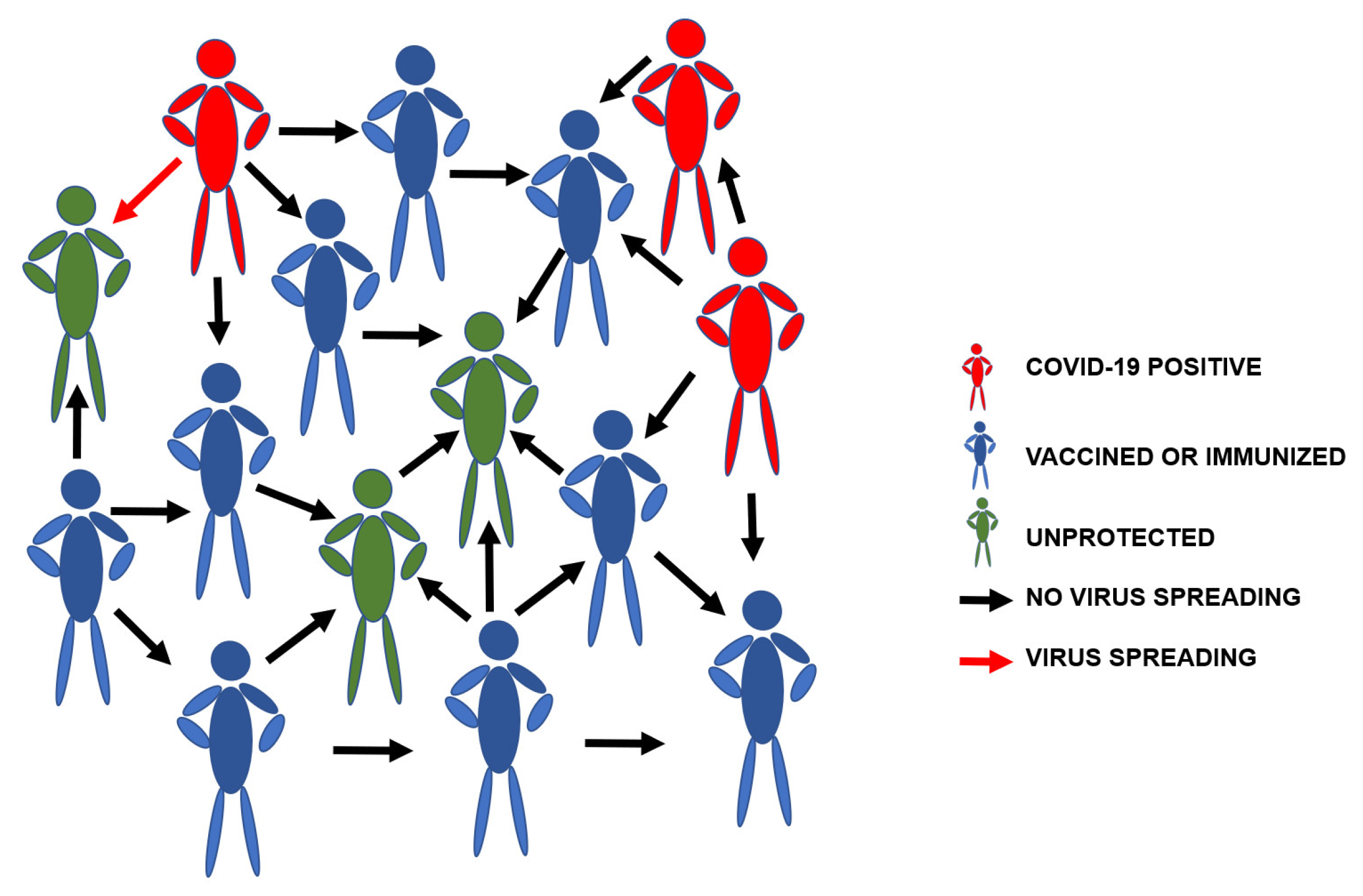
The covid-19 pandemic has halted a lot of what we used to refer to as usual. Daily activities like walking at the park, sharing a seat with a bubbly stranger, or having a good party time at the end of the week are still high in our current to-do lists – once the pandemic is over. The virus has proven not to be a one-period thing and still looks to linger on. However, with the increased research on developing plausible ways to combat this obstacle, the Covid-19 vaccine has been created. Several trials went into the development, with the first trials carried out on July 14th, 2020, for Moderna and a subsequent one on August 12th for Pfizer.
The Current Status of Covid-19 Vaccine Roll-Out
January saw Operation Warp Speed urge Moderna to start rolling out half-dose vaccines to increase the population of those vaccinated, which increased exponentially to 6 million doses by mid-month. The follow-up phases of vaccine enrollments have proved successful and gave birth to UK’s AstraZeneca and Moderna’s Covid -19 vaccines being rolled out as a fit choice. The latest survey shows that President Joe Biden’s adult vaccine coverage is likely to reach a 70% operation estimate by July 4TH. According to the Kaiser Family Foundation’s recent survey, more than 62% of the respondents confirmed to have received the vaccine, rising from 56% in April.
Dr. William Schaffner, a medical director of the National Foundation of Infectious Diseases, was a little surprised by the rising numbers of people taking the vaccine. This he attributed to the possible side effects of the initial vaccines that made people more hesitant. He says that people are gaining confidence but still says that they got to work harder to reach Biden’s threshold. Studies show an increased uptake from Latino adults (57%) and adults without degrees( 55%), but the same cannot be said about the other side of the pie chart. On the bright side, May 10th saw an enrollment of the Pfizer vaccine for children ages 12and older while recording a 40% estimate of adolescents receiving the first dose.
The Current Preventive Measures taken to Combat Future Risks
The Food and Drug Administration has approved an antibody treatment designed to prevent high-risk patients from getting infected. The new drug, sotrovimab, was developed to neutralize the highly infectious virus crippling Brazil, South Africa, and India. Developed by GlaxoSmithKline in the United States, this single drug mimics antibodies that help fight the virus.
It is no doubt that these continuous advances bring us closer to achieving Herd Immunity. A population needs to have 75-80% vaccination to reach the threshold for Herd Immunity, and it is fair to say we are just a mile away from achieving this.
